by Niels Jensen
Precision medicine: treating a patient’s specific cancer based on a genetic understanding of their disease
In precision medicine the basic idea is to develop a treatment for the specific cancer of individual patients based on a genetic understanding of their disease. Precision medicine has also been called personalized medicine or targetted medicine.
The basic idea is not completely new. Already in November 2013 Esquire Magazine reported the development of a treatment specifically for Stephanie Lee’s colon cancer . The treatment effect had been verified on a banana fly, but the board of oncology at Mount Sinai in New York hesitated to give the experimental treatment to the patient and opted for a more conventional treatment. Read the story in Esquire
In Denmark, a collaboration between clinicians at Rigshospitalet and researchers at the Copenhagen University Biotech Research and Innovation Center (BRIC) has systematized a procedure for the development of a precision treatment for each MDS or leukemia patient who is signed up for trial, which has very few exclusion criteria, and straight forward inclusion criteria: you must have either MDS or leukemia and being treated at one of seven hematological centers in Denmark.
From the patient to the lab and back to the patient
When a patient signs up for the trial, they are given a standard protocol treatment depending on the disease and the stage of the disease.
While the patient gets the protocol treatment’ clinicians and researchers get to work: blood and bone marrow samples are collected from the patient. The patient’s tumor cells are then screened in the labs against treatment effect from one or two of more than 400 already commercially available drugs.
If a drug or mixture of drugs shows a positive effect on the patient’s cancer cells, then the second step is initiated. This involved growing the patient’s cancer cells in the lab. Once enough cells are available they are injected into a mouse together with some of the microenvironment from the patient’s bone marrow.
Then the researcher treats the mouse with the drug or drug mixture showed as having a positive effect during the laboratory screening.
If the mouse is successfully treated, then the clinicians have a precision medicine which, in Denmark, can be legally offered to the patient in the clinic.
Doctors in Denmark are allowed to use drugs off-label if they have evidence that the treatment works. This has been done for many years with EPO-like substances to improve red blood cell counts in low risk MDS patients.
Simultaneously, a whole genome next generation sequencing of the samples from the patient is performed to identify the specific mutations in the patient’s cancer cells. This helps researchers to understand why a particular drug or drug mixture have a positive effect, and add the laboratory screening.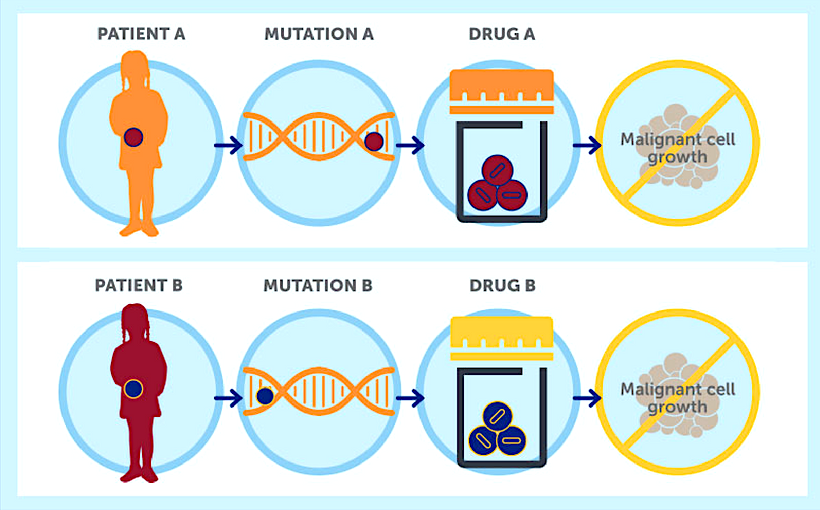
The goal: to optimize the use of already approved drugs and develop novel therapies, in collaboration with pharmaceutical companies
A scientific article published by the University of Copenhagen, Danish Research Center for Precision Medicine of Blood Cancer, explains that the program aims at improving the immediate and long-term outcome for blood cancer patients by coordinating and strengthening ongoing blood cancer research into a program pursuing research questions and integrating results from bench-to-bedside and bedside-to-bench.
The goal is to optimize the use of already approved drugs, identify new targets for therapy, develop novel therapies, test potential novel drugs in pre-clinical models, and collaborate with pharmaceutical companies on developing new drugs and test these and other novel drugs in Phase I-II clinical trials.
Professor Kirsten Grønbæk, PTH Professor and Chief Physician at Rigshospitalet, says:
Via ‘drug screening’ the cancer stem cells from the individual patient could be exposed to 400 different drugs.
In this way, we hope to find the medicine that accurately affects the individual’s cancer stem cells, which is the cause for the cancer to return.
At the same time, we will try to find the molecular changes that indicate that this is the perfect treatment. For some patients, we will immediately find one or more drugs that work, but not for all who are in the need for treatment.
The idea is that while we try to treat a specific patient, we also learn something for the benefit of future patients.
The equipment for this trial have been financed by a grant from the Novo Nordic Foundation. A grant from the Danish Cancer Society covers the expenses for the first three years, and also access to the trial across the country. The trial was conceived by a collaboration between clinician Kirsten Grønbæk, and BRIC researchers Kristian Helin (currently part time at Memorial Sloan Kettering) and Bo Porse.









